It is common knowledge that copper is bad for shrimp, but it’s hard to find actual numbers on that. Let’s dig in.
Tap water contains significant copper
Your water report will tell you how much copper you have in your mains tap water. My water report, which is pretty typical, shows a minimum value for copper of 10 µg/L, a mean of 110 µg/L and a max of 340 µg/L. This variability reflects copper leaching into the water from the copper pipes in your house. The high value of 340 µg/L reflects water that has been sitting in the copper pipes for a while, a so-called “first flush” sample. The minimum of 10 µg/L is what is actually coming in from the mains.
To minimise copper be sure to flush your taps for at least one minute before collecting water for a water change.
The biotic ligand model (BLM) puts actual numbers on copper toxicity to shrimp
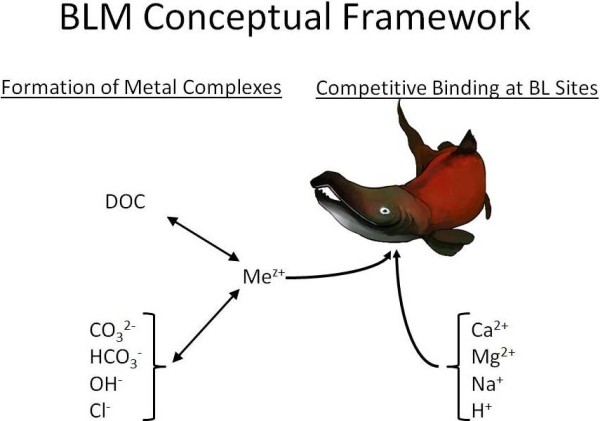
The biotic ligand model was developed specifically to quantify the toxicology from metals like copper, particularly how they interact with and disrupt the biology of gill surfaces in sensitive species like freshwater shrimp. There is a free BLM toxicity calculator you can download to see how your own water stacks up.
The ‘biotic ligand’ in this context is the shrimp’s gill membrane. The essential idea is that anything that can either bind up the toxic metals before they get to the biotic ligand or that can compete with toxic metals for interacting with the biotic ligand will be protective. Negatively charged ions like CO32-, HCO3–, OH– and Cl– can interact with positively charged toxic metals like Cu2+ making the copper less available so are protective. Dissolved organic compounds, particularly humic acids, are quite effective at ‘soaking up’ toxic copper ions and keeping them from interacting with sensitive shrimp tissues. Positively charged ions like calcium, magnesium and sodium can also be protective because they directly compete with the positively charged Cu2+ for interactions with gill membranes.
Using the BLM calculator
You enter the values for these components and the concentration of copper in your water into the calculator and it returns two HQ (hazard quotient) values, one for acute (short-term) toxicity and one for chronic (long-term) toxicity. The HQ values are the ratio of the measured exposure concentration to the threshold concentration at which detrimental health effects are expected to start occurring in sensitive species. Roughly, anything over 1.0 is going to have some harmful consequences, and anything below 1.0 is unlikely to cause problems. The HQ is a sliding and additive scale so the higher above 1.0 you get, the worse the toxicity is going to be and being below 1.0 doesn’t mean totally safe, it just means if that were the only form of toxicity you’d probably be ok, but there might be other environmental stressors you still need to think about.
Shrimphaus BLM results are worrying for copper toxicity
Shrimphaus water is Cambridgeshire tap water that has had alkalinity reduced from 290 mg/L to 15 mg/L. This means Shrimphaus pH is 7.5 but other parameters match mains tapwater: copper is 10 µg/L. DOC is tricky because it is nearly impossible for a hobbyist to measure (needs super-expensive analytical kit) but a conservative value is 2 mg C/L (DOC is measured as the mass of carbon in the organic molecules, not the total mass of those molecules, hence the units mg C/L). Humic acid is similarly non-quantifiable but a conservative estimate is 10%. The other ions that go into the calculator are all read straight off the water report: Ca = 124 mg/L, Mg = 5.71 mg/L, Na = 9.8 mg/L, K = 3 mg/L, SO4 = 32.3 mg/L, Cl = 22.9 mg/L. S (sulfide) is going to be zero – if you can smell the rotten egg smell of hydrogen sulfide you have much bigger problems than copper – but the calculator won’t accept a zero value so put in 0.001 mg/L for that. Punching those numbers into the BLM calculator gives:
- Acute Toxic Units (HQ): 1.02
- Chronic Toxic Units (HQ): 1.64
Oh dear. Acute toxicity is borderline, but chronic toxicity is well above the line. Shrimp are going to be unhappy. Not instantly unhappy, but metabolically stressed over the longer term. I do have a neocaridina population in there that seem relatively healthy and happy and they are breeding successfully, but I also keep caridina in the Shrimphaus, and although they don’t seem to suffer actual accelerated mortality, the caridina pretty much aren’t breeding which is an expected consequence of long-term chronic copper toxicity.
Toxicological hazards are additive and cumulative
Interestingly, if you enter the parameters for actual (not alkalinity reduced) Cambridgeshire tap water into the BLM the copper is not so bad. Full alkalinity tap water here has an equilibrium pH of 8.8 caused by the 290 mg/L alkalinity and the extra HCO3– and CO32- does a pretty good job of binding up the copper. Acute HQ = 0.25 and chronic HQ = 0.40 so the copper is not a problem. However, the high pH is a problem. pH higher than 8.0 starts to dramatically shift the equilibrium between non-toxic ammonium over to the very toxic ammonia and that is even worse for the shrimp than the copper. Tricky stuff. Lowering the pH sorts out the ammonia problem but then uncovers a copper challenge.
Fixing the copper problem
Water conditioner is not going to work
First, let me say definitively:
Do not skip the water conditioner treatment of your water.
The water conditioner is needed to remove chlorine and chloramines. I use Tetra AquaSafe and it’s great for that.
Tetra says AquaSafe also “instantly removes dangerous chlorine, chloramine, copper, zinc and lead from tap water”. Well… sort of… maybe? Tetra doesn’t say how it remove toxic metal ions and the ingredients are secret but the most likely way is with a soluble chelator to bind up those metals and make them unavailable. Probably the chelator is EDTA. EDTA will bind copper, but compared to copper it REALLY binds iron (about a million times better than copper) and there is a pretty healthy 4 µg/L iron in the tap water that will win the ‘bind to EDTA’ battle every time over copper. We don’t know how much EDTA is in AquaSafe, but it can’t be relied upon to take care of the copper problem.
Humic acid can’t be relied upon either
People (including me) like to put botanicals in their shrimp environments. Things like alder cones, catappa leaves and dried oak leaves make for a nice environment for the shrimp. These will release some unknowable amount of humic acids (and a lot of other organic material) which are protective but probably not enough to meaningfully shift the copper toxicity and in any event, like with DOC, by an degree impossible to quantify. If you like using botanicals and your shrimp enjoy them, then that’s great, but it is not a reliably effective copper toxicology management program.
Seachem CupriSorb is the right tool for the job
CupriSorb is designed specifically to remove copper. It is a resin (or more likely a mix of resins), probably based on IDA-functionalised macroporous polystyrene-DVB. Fancy name but this is the classic way to really effectively remove copper and matches the product literature really well.
Looking closely there are relatively uniformly sized clear beads that are a very pale amber colour – these will do the heavy lifting for copper removal – and some more irregular larger white balls with unclear purpose. The copper binding capacity is enormous. 1 g of resin will absorb more than 40 mg of copper. At 10 µg/L copper in the Shrimphaus 12.5 L makes a total of 0.125 mg copper, so 1 g of resin could sort out the copper problem from over 300 Shrimphauses.
The beads are really well behaved in water. They settle rapidly and don’t clump up at all. This is a nice product.
The instructions say to put the CupriSorb in a porous bag (not provided) and to drop that into your filter. Shrimphaus doesn’t have a filter. Hmm…
Treat the water change water with CupriSorb
An easy way to use CupriSorb is to remove the copper from your water before it even gets into the aquarium. The CupriSorb resin beads are small so I spread 1 g of resin between two pieces of fine-textured cut-to-size Juwel PolyPad and inserted that sandwich between the coarse sponges in an EHEIM miniUP pump. Place the pump with the CupriSorb resin in the water you’re going to use for your water change and let that run overnight.
There are a lot of assumptions that go into calculating how much copper will be removed by this whole process but the most conservative estimate is the 10 µg/L copper will be reduced at least 5x down to 2 µg/L, and more likely down to completely negligible levels. The toxicity HQ scales linearly with the amount of copper so even if there are 2 µg/L copper left in the water the acute HQ would be 0.2 and the chronic HQ would be 0.33 so both well comfortably below the safe 1.0 threshold.
The economics of using CupriSorb to polish out residual copper are brilliant. A 250 ml bottle of CupriSorb has 180 g of resin and costs £18 on Amazon. That’s £0.10 per week in resin costs and the bottle will last 3+ years.
The pros use RODI water instead of treatment
If that all sounds like too much work, what the pros do is they remove EVERYTHING from their tap water using a reverse osmosis / deionised (RODI) water system, and then add back the missing KH, GH and trace minerals you need to have. That works brilliantly well. Problems are you have to purchase the RODI kit which starts at £50 minimum and goes up from there and the kit takes up space and needs to be plumbed into your water system. The RODI setup will need filters and resins periodically swapped out. These systems are also somewhat environmentally unfriendly since they typically produce 3 or 4 times as much waste water as the RODI water they make. Still, if you really mean it with shrimp breeding a RODI system is essential.
Shrimphaus water hassle – worth it (?)
As is often the way of things, what started out simple – a low-tech environment for shrimp without any shrimp-eating fish – has built up some considerable complexity over time. Arguably you have to really want it to both strip out the tapwater excess alkalinity with HCl and flow the water through a copper absorbing resin but there you go.
What does local fish store (LFS) do? They sell the less sensitive neocaridina and they keep them in Cambridgeshire tap water, but not for long enough for the toxic effects of either ammonia or copper to show up. But what happens when you get your shrimp home? Probably they are ok for a while but long-term, at least in Cambridgeshire, you’re going to need to take some extra steps to keep them healthy and happy.



